13 Dec Clinical trial for Ebola vaccine developed at UW–Madison underway in Japan
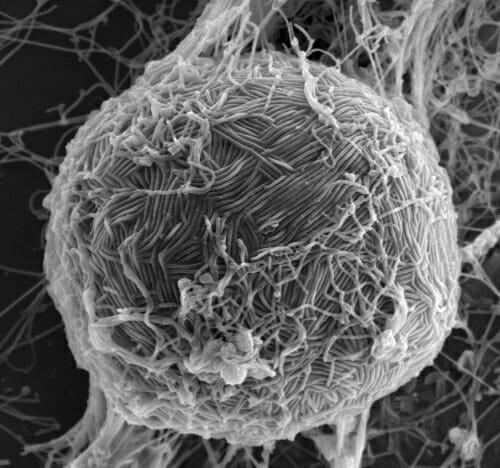
Ebola virus swarms the surface of a host cell in this electron micrograph. Like most viruses, Ebola requires the help of a host cell to survive and replicate. PHOTO: TAKESHI NODA, UNIVERSITY OF TOKYO
As of this week, a phase one clinical trial to test a potential new Ebola vaccine developed by researchers at the University of Wisconsin–Madison is underway in Japan.
Fifteen healthy young men* will receive two doses of the experimental vaccine. If the first group tolerates the vaccine, an additional group of up to 20 volunteers will receive a higher dose of the vaccine.
“In phase one, the main goal is safety,” says Yoshihiro Kawaoka, professor of pathobiological sciences at the UW–Madison School of Veterinary Medicine, who, with Peter Halfmann, a research associate professor in his lab, created the new vaccine.



